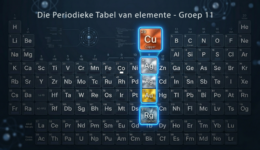
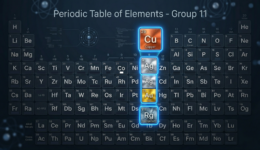
Verkenning van Groep 11 Periodieke Tabel elemente
Sommige elemente is waardevol omdat hulle nuttig is. Ander omdat hulle skaars is.
Groep 11-elemente is albei – en dit is presies waarom mense al duisende jare lank tot hulle aangetrokke is.
Hierdie groep sluit koper, silwer, goud en roentgenium in. Hulle staan dikwels bekend as die muntmetale en het ekonomieë, tegnologieë en kulture op maniere gevorm wat min ander elemente kon. Van antieke munte en juweliersware tot moderne elektronika en hernubare energiestelsels – hul invloed is diepgewortel.
Lank voordat hulle in elektronika of juweliersware gebruik is, het die elemente van Groep 11 reeds die menslike geskiedenis gevorm. Maar wat is die wetenskap agter hul roem? Kyk na “The Atomic Blueprint,”waar ons dieper delf om te sien hoe die unieke struktuur van koper, silwer en goud hulle die uiteindelike geleiers maak.
Wat hulle besonder interessant maak, is hul balans tussen skoonheid en prestasie. Hierdie is nie net aantreklike metale nie – hulle is van die beste geleiers van elektrisiteit en hitte, wat hulle net so belangrik in ingenieurswese maak as in kuns.
Wat definieer Groep 11-elemente?
Op atoomvlak deel Groep 11-elemente ’n soortgelyke struktuur: een elektron in hul buitenste dop, wat bo ’n gevulde stel binneste d-orbitale lê.
Hierdie rangskikking is besonder nuttig. Dit gee hierdie metale hoë elektriese en termiese geleidingsvermoë, terwyl dit hulle ook relatief stabiel en minder reaktief maak as die meeste oorgangsmetale.
Hulle vorm tipies ’n +1 oksidasietoestand, hoewel koper en goud onder die regte omstandighede verder kan gaan. Oor die algemeen reageer hulle egter nie aggressief met hul omgewing nie – en dit is deel van hul aantrekkingskrag.
Dit is waarom goud vir eeue onveranderd kan bly, en waarom koperbedrading vir dekades kan hou.
’n Groep gevorm deur subtiele tendense
Soos jy afbeweeg in Groep 11, is die veranderinge geleidelik maar betekenisvol.
Atome word groter en swaarder, digtheid neem toe, en weerstand teen korrosie verbeter. Reaktiwiteit neem oor die algemeen af en bereik sy laagste punt by goud, een van die minste reaktiewe metale wat bekend is.
Hierdie is nie dramatiese veranderinge nie – maar dit maak saak. Dit verduidelik waarom koper wyd in infrastruktuur gebruik word, terwyl goud gereserveer word vir toepassings waar stabiliteit krities is.
Koper: Die metaal wat die moderne wêreld gebou het
Koper is waar praktiese bruikbaarheid en prestasie bymekaar kom.
Dit word al meer as 10 000 jaar deur mense gebruik en is vandag steeds een van die belangrikste industriële metale. Die rede is eenvoudig: dit gelei elektrisiteit uitstekend, dit is duursaam, en dit is relatief volop.

Daardie kombinasie maak dit die ruggraat van moderne infrastruktuur. Elektriese bedrading, kragnetwerke, elektronika, loodgieterstelsels – koper kom in alles voor.
Met verloop van tyd reageer dit stadig met lug om ’n groen oppervlaktelaag te vorm wat as ’n patina bekend staan. In plaas daarvan om die metaal te beskadig, beskerm hierdie laag dit teen verdere korrosie.
Dit is een van daardie seldsame materiale wat met ouderdom verbeter.
Silwer: Ongeëwenaarde geleidingsvermoë
As koper uitstekend is, is silwer uitsonderlik.
Dit is die beste geleier van elektrisiteit van enige element. Dit weerkaats ook lig uiters goed en dra hitte doeltreffend oor. Op suiwer prestasievlak oortref silwer elke ander metaal in sy groep.

Waarom word alles dan nie van silwer gemaak nie?
Koste.
Omdat dit duurder is, word silwer selektief gebruik – hoofsaaklik in hoëprestasie-elektronika, sonpanele en gespesialiseerde bedekkings. Dit kom ook voor in juweliersware en dekoratiewe items, en behou sy lang verbintenis met rykdom en vakmanskap.
Silwer verkleur wel met tyd deurdat dit met swael in die lug reageer. Dit is egter slegs ’n oppervlakeffek en beïnvloed nie die geleidingsvermoë daarvan nie.
Goud: Stabiliteit bo alles
Goud volg ’n ander pad.
Waar koper en silwer ’n balans tussen prestasie en koste bied, fokus goud volledig op stabiliteit. Dit is uiters onreaktief – dit verkleur nie, korrodeer nie en versleg nie onder normale omstandighede nie.

Dit is waarom goudvoorwerpe duisende jare kan oorleef sonder om hul glans te verloor.
Maar goud is nie net vir versiering nie. Sy betroubaarheid maak dit noodsaaklik in elektronika, veral in verbindings en stroombane waar mislukking nie ’n opsie is nie. Dit word ook in lugvaart, medisyne en presisie-toerusting gebruik.
’n Verdere kenmerkende eienskap is sy smeebaarheid. Goud kan tot ’n uiterste mate gerek en gevorm word sonder om te breek, wat dit een van die mees bewerkbare metale maak wat bekend is.
Roentgenium: Die teoretiese lid
Heel onder in die groep is roentgenium, element 111.
Anders as die ander kom dit nie natuurlik voor nie. Dit word in deeltjieversnellers geskep en bestaan slegs vir breuke van ’n sekonde voordat dit verval.

Omdat hiervan word sy eienskappe meestal voorspel. Wetenskaplikes verwag dat dit soortgelyk aan goud sal optree, maar direkte waarneming is baie beperk.
Vir nou gaan roentgenium minder oor praktiese gebruik en meer oor die uitbreiding van ons begrip van atoomstruktuur.
Fisiese eienskappe: Gebou vir prestasie
Groep 11-metale deel ’n stel fisiese eienskappe wat hul langtermyn-belangrikheid verklaar.
Hulle gelei elektrisiteit en hitte uiters goed, wat hulle sentraal maak in kragstelsels en elektronika. Hulle is ook smeebaar en rekbaar, wat beteken dat hulle gevorm en uitgerek kan word sonder om te breek.
Hul kenmerkende metaalglans maak hulle gewild vir dekoratiewe gebruik, terwyl hul digtheid – veral by goud – bydra tot hul duursaamheid en waarde.
Hierdie eienskappe maak hulle nie net nuttig nie – dit maak hulle betroubaar.
Chemiese gedrag en reaktiwiteit
In vergelyking met ander oorgangsmetale is Groep 11-elemente relatief onreaktief.
Koper reageer stadig met suurstof en vorm met verloop van tyd sy bekende groen laag. Silwer reageer met swaelverbindings, wat verkleuring veroorsaak. Goud reageer egter byna glad nie – dit weerstaan bykans alles, behalwe baie aggressiewe chemiese mengsels soos aqua regia.
Hierdie lae reaktiwiteit is ’n groot voordeel. Dit laat hierdie metale toe om hul eienskappe oor lang tydperke te behou, selfs in uitdagende omgewings.
Waar hierdie elemente voorkom
In die natuur kom hierdie metale in verskillende vorme voor.
Koper kom wyd in ertse voor en word op groot skaal ontgin. Silwer word in ertse sowel as in sy natuurlike metaalvorm aangetref. Goud is bekend daarvoor dat dit natuurlik in suiwer vorm voorkom, dikwels in riviere of in rots ingesluit.
Roentgenium, daarenteen, kom glad nie natuurlik voor nie.
Die beskikbaarheid van koper maak dit geskik vir wydverspreide gebruik, terwyl die skaarsheid van silwer en goud bydra tot hul waarde.
Waarom Groep 11 belangrik is
Groep 11-elemente het nog nooit hul relevansie verloor nie – hulle het net aangepas.
Hulle het begin as materiale vir munte en ornamente, simbole van rykdom en mag. Vandag is hulle sentraal tot elektriese stelsels, hernubare energietegnologie en gevorderde elektronika.
Koper hou die wêreld verbind. Silwer stoot prestasie verder. Goud verseker betroubaarheid waar dit die meeste saak maak.
Saam wys hulle hoe een groep in die Periodieke Tabel antieke geskiedenis en moderne innovasie kan verbind – sonder om hul belangrikheid te verloor.
Vrae wat gereeld gevra word
Watter elemente is in Groep 11?
Koper, silwer, goud en roentgenium
Waarom word hulle muntmetale genoem?
Omdat koper, silwer en goud histories gebruik is om munte te maak.
Watter een is die beste geleier?
Silwer gelei elektrisiteit beter as enige ander element.
Waarom verkleur goud nie?
Omdat dit baie onreaktief is en nie maklik verbindings met suurstof of ander stowwe vorm nie.