There’s something quietly magical about a classroom moment when learners lean in, eyes wide, and say, “Wait… do that again.”
The humble pepper-and-water activity has exactly that effect. It’s simple, a little theatrical, and – most importantly – packed with scientific meaning.
As we approach World Hand Hygiene Day on 5 May, it offers South African Physical Science or Natural Sciences teachers a perfect opportunity to connect curriculum concepts with real-world relevance.
The ‘Pepper and Water’ demo becomes a vivid story about how Chemistry protects our communities when viewed through the lens of World Hand Hygiene Day. Click here to watch ‘The Chemistry of the Bowl’ and discover how to use this Doc Scientia practical to connect surface tension concepts with the real-world power of soap.”
At first glance, the setup couldn’t be easier. A shallow bowl, some water, a sprinkle of black pepper. The pepper rests lightly on the surface, almost as if it’s been carefully placed there one grain at a time.
Ask a learner to dip a clean finger into the centre and… nothing much happens. Maybe a few flakes cling to their skin, but the surface remains mostly undisturbed.
Then comes the twist.
Add a drop of dish soap to the finger and try again. Instantly, the pepper shoots outward, racing to the edges of the bowl as though it’s been startled. A clear space opens in the middle, and just like that, your classroom has gone from mildly curious to fully engaged.
What’s powerful about this demonstration is not just the visual drama, but the layers of science sitting beneath it. On one level, it’s a beautiful illustration of surface tension. Water molecules cling to one another through cohesion, forming a kind of invisible “skin” on the surface. This is strong enough to support the pepper, which, being hydrophobic, doesn’t mix with the water and simply floats.
Introduce soap, however, and that delicate balance is disrupted. Soap molecules are surfactants – surface-active agents designed to interfere with those cohesive forces. As the soap spreads, it breaks the hydrogen bonds between water molecules. The water, in response, pulls away from the soap, dragging the pepper along for the ride.
What learners are seeing is not just movement, but a molecular tug-of-war playing out in real time.
And this is where the practical activity becomes more than just a Physics or Chemistry demo – it becomes a story about hygiene.
In the context of handwashing, soap plays a far more critical role than simply “making things clean.” Many viruses, including those learners have become all too familiar with in recent years, are surrounded by a lipid bilayer – a fatty envelope that protects them. Soap molecules, with their hydrophilic heads and lipophilic tails, are perfectly designed to break this structure apart. The tails wedge themselves into the lipid layer, destabilising it, and ultimately tearing the virus open.
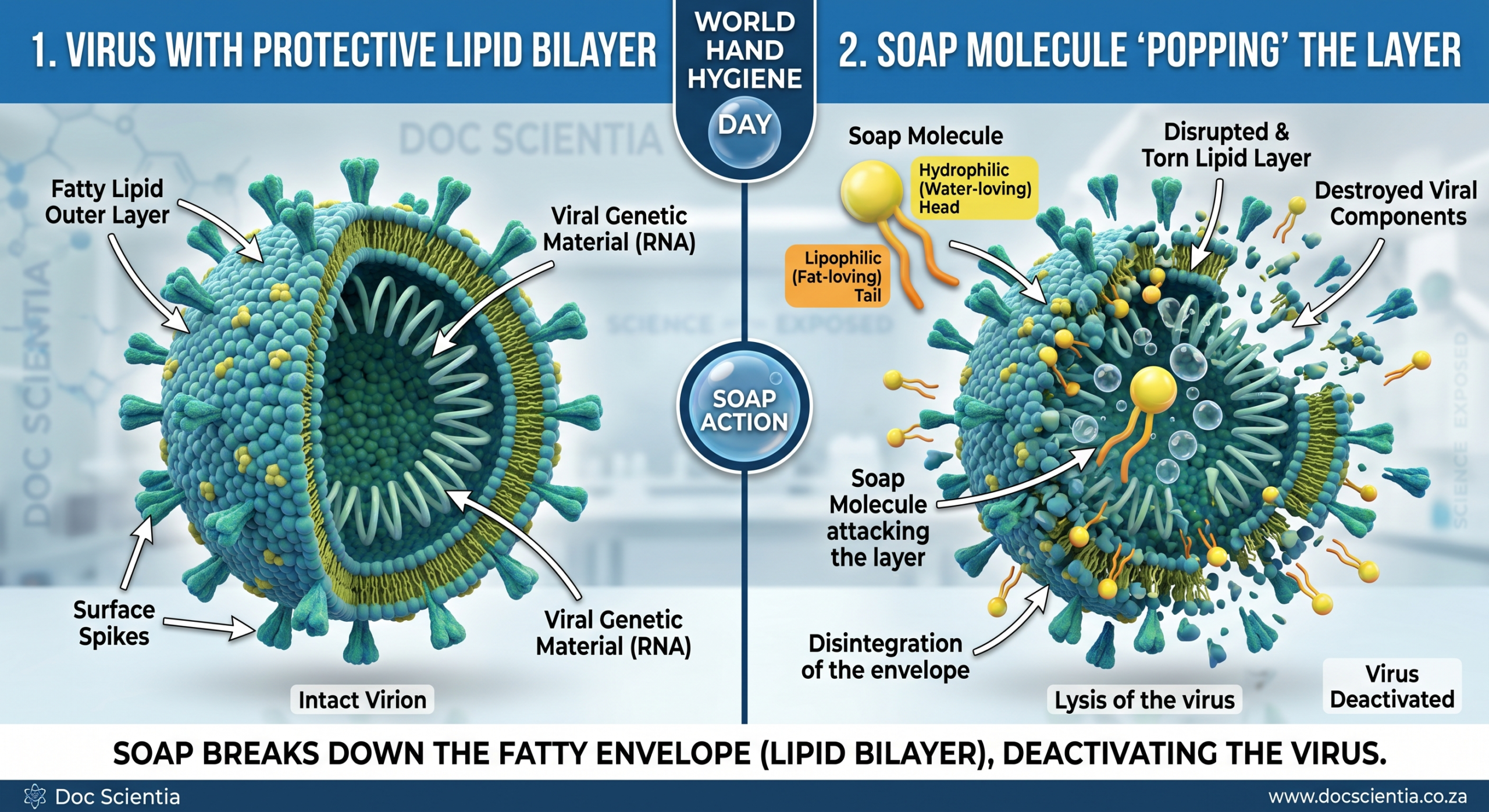
Of course, the pepper doesn’t represent germs in a literal sense. It doesn’t dissolve or get destroyed. But as a metaphor, it’s incredibly effective. Learners can see that soap doesn’t just passively rinse things away – it actively disrupts the environment.
For South African classrooms, where practical demonstrations can sometimes be limited by resources, this practical activity is a gift. It’s low-cost, quick to set up, and adaptable across different phases. Whether you’re introducing intermolecular forces in Physical Sciences or exploring basic hygiene concepts in Natural Sciences, it slots in seamlessly.
A useful tip: lean into the storytelling. Tell your learners the pepper represents germs, and ask them to predict what will happen before you add the soap. Let them be surprised. That moment of surprise is often where the real learning begins.
As World Hand Hygiene Day approaches, this small practical activity offers a big message. Science isn’t just something that happens in textbooks or laboratories – it’s in the everyday actions we sometimes take for granted. And sometimes, all it takes is a bowl of water, a pinch of pepper, and a drop of soap to make that clear.
Practical Activity
Aim: To test if soap has the power to physically manipulate and disrupt the environment where germs live.
Apparatus:
- Shallow bowl or plate
- Water
- Black pepper
- Dishwashing soap
Method:
- The Setup: Fill a shallow bowl or plate with water and sprinkle a layer of black pepper across the surface. The pepper floats because it is light and the surface tension of the water holds it up.
- The Touch: Dip a clean finger into the centre. Nothing happens – the pepper stays put (and might even stick to your finger).
- The Magic: Rub a small drop of dishwashing soap on your finger and touch the centre of the water again.
Observation:
The pepper instantly “races” to the edges of the plate, leaving a clear circle of water in the middle.
The Science (The “Why”)
There are actually two scientific principles at play here that Doc Scientia learners would find fascinating:
- Surface Tension (Physical Sciences)
Water molecules are very attracted to one another (cohesion). On the surface, they cling together to form a “skin.” Because pepper is hydrophobic (water-fearing), the water doesn’t wet it, and the surface tension allows the flakes to float.
- The Role of the Surface-active substance (Chemistry)
Soap is a surface-active agent. Its primary job is to break the surface tension of the water.
- As the soap moves through the water, it breaks the hydrogen bonds between water molecules.
- The water molecules want to keep their surface tension intact, so they pull away from the soap, carrying the pepper flakes with them to the edges of the bowl.
Why use this for Hand Hygiene Day?
While the pepper practical activity shows water moving away, it visually demonstrates how soap breaks a barrier.
In the case of a virus, that “barrier” is the lipid bilayer. Just like the soap forces its way into the water surface, the lipophilic (fat-loving) tails of soap molecules force their way into the fatty envelope of a virus and tear it open.