Key highlights
- Transition metals are a large group of elements located in the d-block of the Periodic Table.
- These metals, identified by their atomic number, exhibit unique physical properties like high melting points and densities.
- They have various important uses, from industrial catalysts to components in electronic devices.
- Transition metals are distinct from rare earth elements, though they share some similar properties.
- Elements like scandium and yttrium are often classified as a transition metal and a rare earth element.
- Their discovery and classification have evolved over time with advances in science and technology.
If you’ve ever glanced at the Periodic Table and paused at the big block in the centre, you’ve already met the transition metals – you just might not have known their name.
These elements are everywhere. They’re in the steel holding up buildings, the copper running through your walls, and the tiny components powering your phone. Quietly, they’re doing most of the heavy lifting in modern life.
What makes them so useful comes down to something you can’t see: the way their electrons are arranged. That structure gives them unusual flexibility – letting them form strong alloys, act as catalysts, and take part in reactions other elements simply can’t handle.
In this guide, we’ll break down what transition metals are, how they’re organised in the Periodic Table (especially the often-debated Group 3), and why they matter so much in industries ranging from construction to clean energy.
What makes transition metals different?
At a glance, transition metals might just look like another group of elements – but chemically, they behave very differently from the rest of the Periodic Table.
The key lies in their electron structure. Transition metals have partially filled d-orbitals, which gives them a kind of built-in flexibility. Unlike elements that stick to one stable form, these metals can shift between different oxidation states depending on the reaction they’re involved in.
That’s a big deal.
It’s what allows iron to form both Fe²⁺ and Fe³⁺ ions. It’s why many transition metals create vividly coloured compounds. And it’s what makes them incredibly effective as catalysts – speeding up chemical reactions without being consumed in the process.
You can see the effects of this everywhere:
- Iron in construction
- Platinum in catalytic converters
- Copper in electrical wiring
This same flexibility also explains why some elements – like scandium and yttrium – don’t fit neatly into one category. They sit in the transition metals group but behave a lot like rare earth elements, which is where things start to get interesting (and a bit messy).
In short, transition metals aren’t just defined by where they sit on the Periodic Table – they’re defined by how adaptable they are.

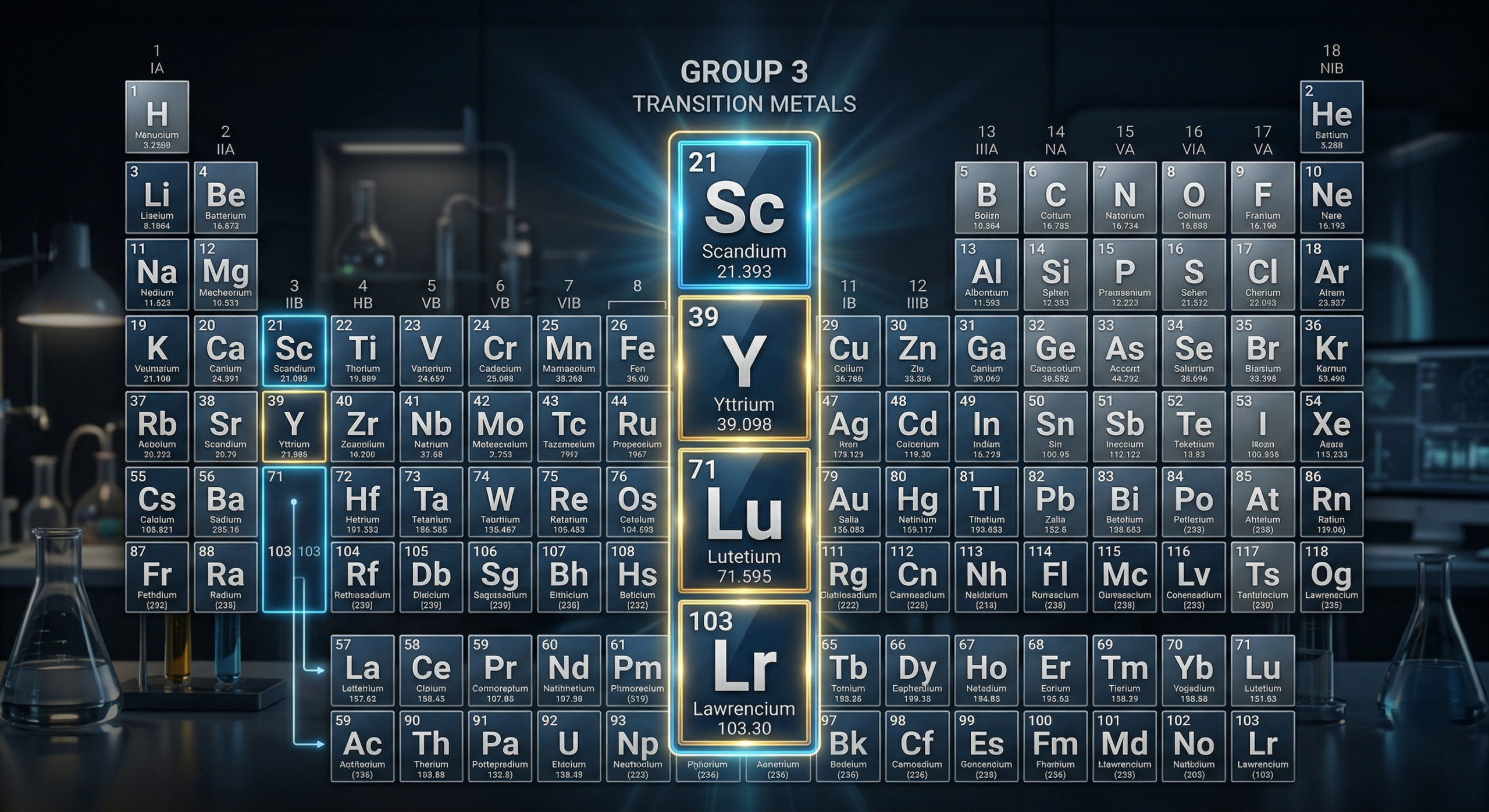
Group 3 elements: Where things get complicated
Most of the Periodic Table is tidy and predictable. Group 3 is not.
This small set of elements – scandium (Sc), yttrium (Y), and either lutetium (Lu) or lanthanum (La), depending on who you ask – sits right at the boundary between the transition metals and the rare earth elements. And that’s where the confusion begins.
Scandium and yttrium are firmly placed in the d-block, which makes them transition metals by definition. But chemically, they behave a lot like rare earth elements. They’re typically found in the same mineral deposits, and almost always form ions with a +3 oxidation state.
So which are they, really?
The honest answer: a bit of both.
Why Group 3 doesn’t fit neatly
Most transition metals are known for their flexibility – they can adopt multiple oxidation states and participate in a wide range of reactions. Group 3 elements are different.
They’re much more consistent.
Scandium and yttrium almost exclusively form +3 ions. That puts them chemically closer to the lanthanides (the rare earth elements), which also stick to a +3 state in most situations.
This overlap is why you’ll often see scandium and yttrium included in discussions about rare earths, even though they technically belong to the transition metals.
The lutetium vs lanthanum debate
Things get even more interesting when you look at the third slot in Group 3.
Some Periodic Tables list:
- Scandium (Sc)
- Yttrium (Y)
- Lanthanum (La)
- Actinium (Ac)
Others prefer:
- Scandium (Sc)
- Yttrium (Y)
- Lutetium (Lu)
- Lawrencium (Lr)
The disagreement comes down to how you prioritise electron configuration versus chemical behaviour. Lutetium fits more cleanly with the d-block structure, while lanthanum aligns better with historical and chemical trends.
There isn’t universal agreement, and that’s part of what makes Group 3 so interesting – it’s one of the few places where the Periodic Table still sparks real debate.
A bridge between two worlds
In many ways, Group 3 acts as a bridge.
It connects the d-block transition metals with the f-block rare earth elements, both structurally and chemically. This is why elements like scandium feel like they belong to two families at once.
And while that might seem like a technical detail, it actually matters. This overlap influences how these elements are mined, classified, and used in modern technologies – from aerospace alloys to advanced electronics.
So if the Periodic Table looks clean and orderly at first glance, Group 3 is a reminder that nature doesn’t always follow neat categories.

Rare earth elements vs. transition metals: What’s the real
difference?
It’s easy to mix up rare earth elements and transition metals – especially since elements like scandium and yttrium seem to belong to both groups. But the core difference between them is surprisingly simple. It all comes down to where their electrons sit.
Transition metals fill their d-orbitals. Rare earth elements fill their f-orbitals. That small shift in electron structure leads to big differences in how they behave.
Transition metals are versatile. They can adopt multiple oxidation states, form a wide variety of compounds, and often act as catalysts in chemical reactions. This is why elements like iron, copper, and platinum are so widely used across industries.
Rare earth elements, on the other hand, are much more consistent. Most of them prefer a +3 oxidation state and behave very similarly to one another. That similarity is actually a challenge – it makes them difficult to separate during extraction, which is a big part of why they’re considered “rare.”
Why the confusion exists
The overlap mostly comes from scandium and yttrium.
They are technically transition metals based on their position in the Periodic Table. But chemically, they behave like rare earth elements – and they’re often found in the same mineral deposits.
Because of this, they’re frequently grouped with rare earths in industrial and economic contexts, even if that’s not strictly accurate from a chemistry standpoint.
A practical way to think about it
If you want a quick way to keep the two groups straight:
- Transition metals are chemically flexible and widely used in structural materials,
electronics, and catalysts - Rare earth elements are chemically similar to each other and critical for high-tech
applications like magnets, lasers, and clean energy systems
Both groups are essential – but for very different reasons.
Understanding that difference makes the Periodic Table feel a lot less like a chart to memorise, and more like a system that actually explains how the material world works.
If you’re struggling to visualise how these elements differ, watch the Group 3 Transition Metals explanation video here.
Some of scandium’s main applications include:

- Aerospace: Used in high-performance components for military aircraft, like the MiG-21 and MiG-29.
- Sports Equipment: Found in lightweight, high-strength bicycle frames, baseball bats, and lacrosse sticks.
- High-Intensity Lighting: Scandium iodide is used in metal-halide lamps to produce a light source that resembles natural sunlight.
- Fuel Cells: Scandium oxide is used to stabilise zirconia in solid oxide fuel cells, improving their efficiency and durability in clean energy technologies.
Yttrium (Y): Properties and practical roles

The properties of yttrium compounds are intermediate between those of aluminum and scandium. It is a key ingredient in various advanced materials and technologies due to its unique characteristics. Its compounds are used as phosphors, giving the red color in old CRT television screens.
Yttrium has several important practical roles, including:
Lasers: Yttrium-aluminum garnet (YAG) is a widely used crystal in lasers.
Medical Technology: Yttrium oxide is used to stabilise zirconia in medical implants and is found in some cancer treatments. It is not directly used in MRI machines, but other rare earths are.
Camera Lenses: Yttrium oxide can be used to make high-quality, shock- resistant camera lenses.
Superconductors: It was a component in the first high-temperature superconductors ever discovered.
Lawrencium (Lr): Properties and practical roles

Lawrencium has no known practical, industrial, or biological roles outside of scientific research. Its significance is purely academic.
Nuclear Physics Research: Used to study the properties of superheavy elements, test models of nuclear stability, and define the limits of the periodic table.
Relativistic Chemistry Studies: Its anomalous electron configuration makes it a key subject for testing relativistic quantum chemistry theories.
Isotope Studies: Research focuses on isotopes 256Lr (27 s) and 260Lr (2.7 min) for chemical experiments.
South Africa’s role in transition metal production
South Africa holds a uniquely important position in the global production of several key transition metals. The nation’s wealth of natural resources is concentrated in the Bushveld Igneous Complex, a massive geological formation that contains some of the richest ore deposits on the planet.
This region is the world’s largest source of platinum-group metals (PGMs), including platinum, palladium, and rhodium. These metals are critical for catalytic converters in vehicles. South Africa is also a leading producer of chromium, which is essential for making stainless steel, and manganese, another key component in steel production.
The country’s ability to mine and process these valuable resources makes it a crucial player in the global supply chain for many industries. South Africa's contribution ensures the steady availability of these essential transition metal elements, which are foundational to modern manufacturing and technology.
Key mining techniques
A variety of mining techniques are used to extract transition metals and rare earth elements from the ground. The method chosen depends on the type of deposit, its depth, and the concentration of the desired metal. Open-pit mining is common for large, near-surface deposits, while underground mining is used for deeper ore bodies.
Once the ore is mined, the extraction process begins. This typically involves crushing and grinding the rock, followed by chemical processing to separate the valuable metals. Techniques like froth flotation, leaching with acids, and electrowinning are used to isolate and purify the metals.
These mining and extraction processes can have significant environmental impacts. Issues like water pollution from chemical runoff, habitat destruction, and the generation of large amounts of waste rock (tailings) are major concerns. Furthermore, improper disposal of electronic waste can lead to hazardous materials leaching into the
environment, highlighting the need for sustainable practices and recycling.
Abundance in earth’s crust
The abundance of transition metals in the Earth’s crust varies widely. Iron is the fourth most abundant element overall, making up about 5% of the crust. This makes it readily available and relatively inexpensive. Other transition metals like titanium and manganese are also quite common.
In contrast, some transition metals like gold and platinum are extremely rare, which contributes to their high value. The concentration of an element is just as important as its overall abundance. For a deposit to be economically viable, the metal must be concentrated enough to make extraction worthwhile.
Interestingly, many rare earth elements are more abundant in the Earth’s crust than some transition metals. For example, cerium is more common than copper. The “rarity” of REEs comes from their dispersion and the difficulty of separating them from each other, not their lack of presence in the crust.
Extraction and processing
Getting transition metals and rare earth elements from raw ore into a usable form is a complex, multi-step process. The journey involves extraction from the rock, followed by extensive refinement and purification to achieve the desired purity. Each step requires specialised chemical and engineering techniques.
These processes are not without their challenges, including significant environmental considerations. Let’s explore the techniques used for extraction and processing, and the environmental impact that comes with them.
Techniques for extraction
The extraction of transition metals and rare earth elements from their ores is a chemically intensive process. After mining and crushing the rock, the minerals are typically subjected to a process called leaching. This involves using strong chemicals, often acids like sulfuric acid or hydrochloric acid, to dissolve the desired metals out of
the ore, creating a chemical solution.
For rare earth minerals, this step is particularly challenging because all the REEs tend to dissolve together. Once the metals are in solution, they must be separated from other impurities and, in the case of REEs, from each other.
To produce metallic scandium, for instance, scandium oxide is first converted to scandium fluoride. This compound is then reduced using metallic calcium in a high- temperature reaction to produce pure scandium metal. Different transition metals require different chemical pathways, but the general principle of chemical separation and reduction is common.
Refinement and purification
After initial extraction, the metals are still in a relatively impure state and must undergo refinement and purification. This is a critical step to produce metals with the specific properties required for high-tech applications. The methods used depend on the metal and the level of purity needed.
For many transition metals, processes like electrolysis or fractional distillation are used. In electrolysis, an electric current is passed through a solution containing the metal ions, causing the pure metal to deposit on an electrode. This is a common method for purifying copper.
The refinement of rare earth elements is particularly difficult due to their chemical similarity. The process often involves complex and repetitive techniques like solvent extraction or ion exchange chromatography. These methods exploit subtle differences in the properties of the REE ions to gradually separate them, one by one. This lengthy
and costly purification process is a major reason for the high price of individual REEs.
Environmental considerations
The extraction and processing of transition metals and rare earth minerals come with significant environmental considerations. The extensive use of toxic chemicals, such as strong acids and organic solvents, can lead to severe water pollution if not managed properly. These substances can contaminate local water sources, harming ecosystems and human health.
Mining operations also generate massive amounts of waste, including tailings (the leftover rock after extraction) which can contain radioactive materials, particularly in rare earth mining. The disposal of this waste is a major challenge. Furthermore, the energy- intensive nature of these processes contributes to greenhouse gas emissions.
The growing mountain of electronic waste adds another layer to the problem. Discarded electronics contain valuable metals, but improper disposal releases hazardous substances into the environment. Promoting recycling and developing more sustainable extraction methods are crucial steps to mitigate these environmental issues.
South African case studies
South Africa is the world’s largest resource of platinum-group metals (PGMs), which are essential for catalytic converters that reduce harmful emissions from vehicles. The extraction processes used here are highly sophisticated, tailored to the unique geology of the region.
The mining industry in South Africa has had to innovate to overcome challenges, including deep ore bodies and complex mineralogy. Companies have developed specialised smelting and refining techniques to separate the various PGMs – platinum, palladium, rhodium, and others – from each other and from other base metals like nickel
and copper.
These metals are not only vital for the automotive industry but also for emerging clean energy technologies. Platinum, for example, is a key catalyst in hydrogen fuel cells.
South Africa&’s role as a primary supplier of these critical transition metals positions it at the forefront of both traditional and future-facing industries.
Conclusion
Transition metals sit at the centre of the Periodic Table – and, in many ways, at the centre of modern life. Their unique electron structures give them a level of flexibility that few other elements can match, which is why they show up everywhere from construction materials to cutting-edge technologies.
Group 3 highlights just how complex this part of the Periodic Table can be. Elements like scandium and yttrium don’t fit neatly into one category, blurring the line between transition metals and rare earth elements. That overlap isn’t just a technical detail – it shapes how these materials are classified, sourced, and used in real-world applications.
And while rare earth elements aren’t truly “rare,” they are difficult to extract and refine, making them strategically important – especially as demand grows for clean energy technologies, electronics, and advanced manufacturing.
Taken together, these elements tell a bigger story. The Periodic Table isn’t just a chart – it’s a map of how materials behave, interact, and ultimately power the world around us.
Understanding transition metals is one small step toward understanding that much larger system.
Frequently asked questions
Are rare earth elements really rare in the Earth’s crust?
No, most rare earth elements are not geologically rare in the Earth's crust. For example, cerium is more abundant than copper. The term “rare” refers to the fact that these rare metals are seldom found in economically mineable concentrations and are difficult to separate from one another.
How are transition metals used in modern technology?
Transition metals are vital in modern technology. They are used in electronics (copper wiring), alloys (steel), clean energy technologies like wind turbines and fuel cells, and medical equipment such as MRI machines. Their unique properties make them essential for countless advanced applications.
What is the difference between transition metals and rare earth elements?
The main difference lies in their electron configurations. Transition metals fill their d- orbitals, while rare earth elements fill their f-orbitals. This leads to different chemical behaviours, though some elements like scandium and yttrium share similar properties and are often grouped with both.




